Historia corta
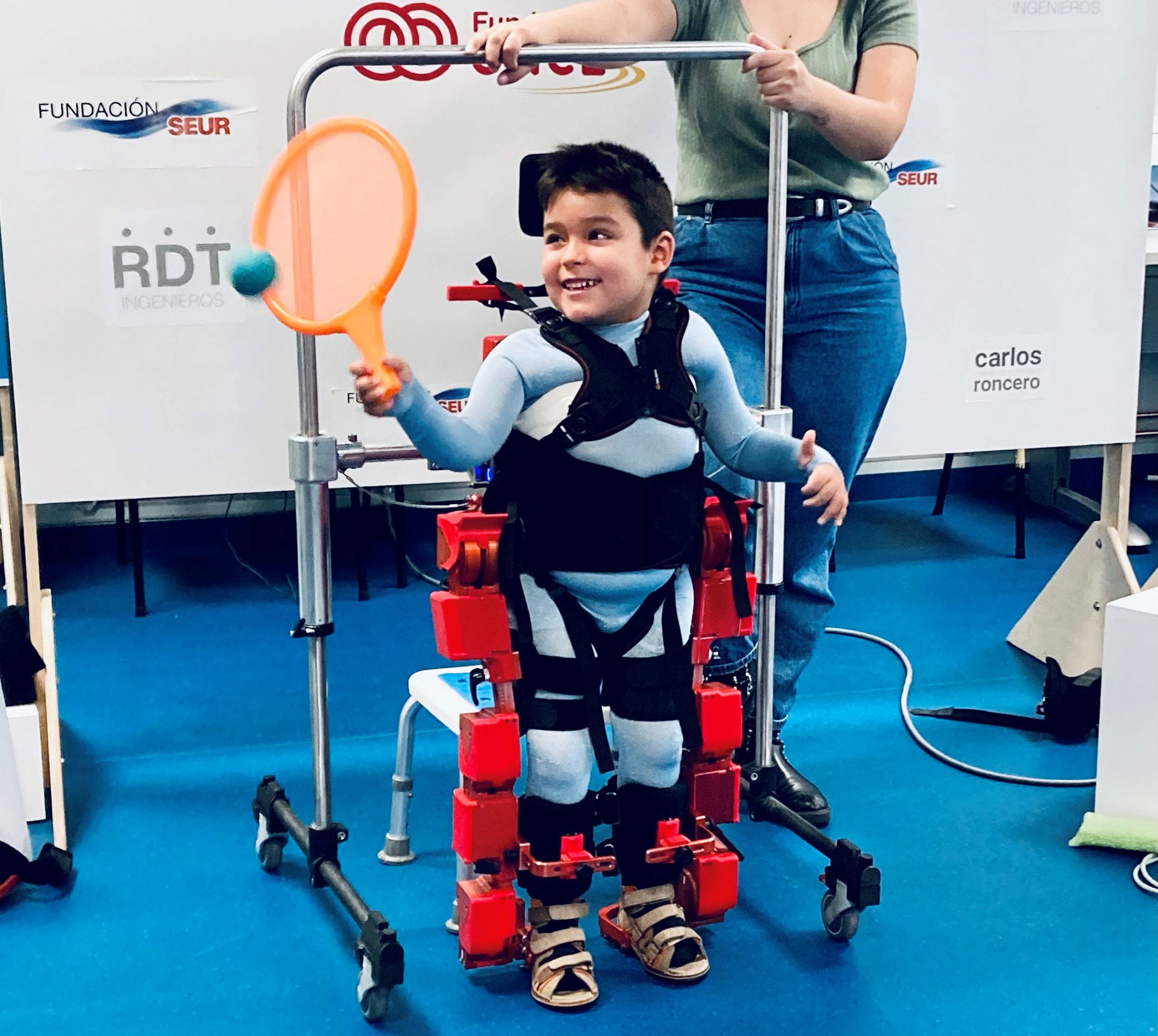
Alvaro tiene 11 años. Cuando nació era tan rubio que las pestañas y las cejas eran transparentes. Para nosotros era precioso, perfecto. Un día, cuando tenía seis meses, observamos que sus movimientos empezaban a ser más torpes. Acudimos a diferentes pediatras y todos decían que nuestro hijo era “vago” y por eso no gateaba, no podía apenas comer y por eso sus manos temblaban. Tardamos seis meses en encontrar una pediatra que nada más ver al niño reconoció que esos temblores de manos eran daño neurológico, y nos envió al hospital. El diagnóstico genético nos confirmó que tenía AME, y las palabras con las que describieron esta enfermedad fueron “enfermedad degenerativa e incurable”. Estas son palabras que espero que ningún padre tenga que oír jamás. No nos rendimos, buscamos terapias y especialistas que apostasen por nuestro hijo. Con mucha fisioterapia diaria, médicos que lucharon por él y gracias sobre todo a la gran fuerza interior que Álvaro tiene, superamos los dos años que le daban de vida, y cuando tenía casi tres años, conocimos el proyecto de Marsi Bionics y el CSIC: El Exoesqueleto Pediátrico. Un invento futurista que lograba lo que absolutamente todos los especialistas a nivel mundial en AME aseguraban que nunca haría un niño como Álvaro: Andar. El uso de este maravilloso invento puede significar para Álvaro y otros niños como él vivir más años con mayor calidad de vida.
Crowdfunding Álvaro 11 años / Atrofia Muscular Espinal (2022)
7.000,00€
Objetivo de financiación-
1.865,76€
Fondos recaudados -
La campaña no termina nunca
Método de fin de campaña
Historia de la campaña
Durante el año 2021 el objetivo de la financiación fue de 31.770,00€, se logró recaudar 25.183,00€.
| Nombre | Cantidad de la donación | Fecha |
|---|---|---|
| Minguela | 20,00€ | May 30, 2023 |
| Cynthia Jiménez | 30,00€ | December 12, 2022 |
| Amor de la Riera | 10,00€ | November 16, 2022 |
| Clinica Oseo | 90,00€ | October 12, 2022 |
| R+J | 300,00€ | October 10, 2022 |
| VK | 100,00€ | April 05, 2022 |
| Yelena | 100,00€ | February 22, 2022 |
| Remanente de donaciones a 1 de Enero 2022 | 1.205,76€ | February 14, 2022 |
| Fernando | 10,00€ | February 13, 2022 |